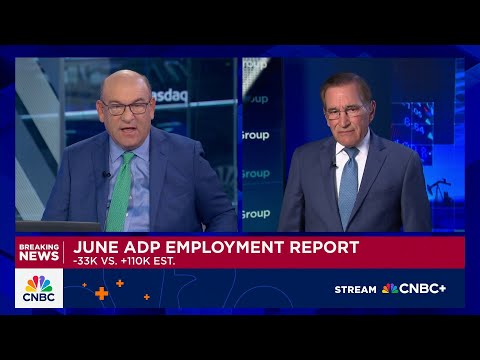
Strong and Weak Acids - Examples and Explanation
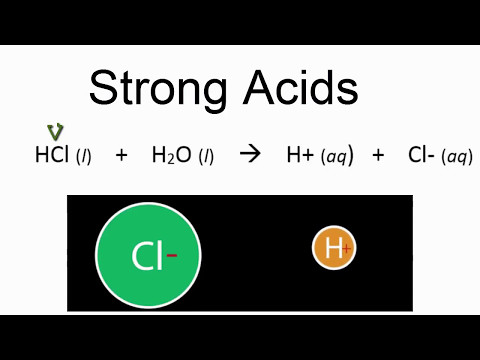
The primary difference between strong and weak acids is how the behave in water. Strong acids dissociate (break apart) completely in water). For example, HCl, a strong acid will break apart into H+ and Cl- ions. Weak acids dissociate partially in water. For example, HF, a weak acid, will only have some of the HF molecules dissociated at any ...