Strong vs. Weak Bases - What's the difference? How do they dissociate? The 8 Strong Bases
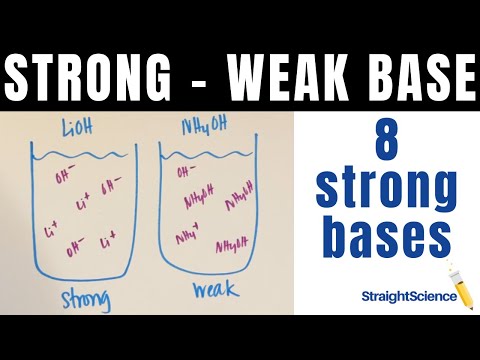
In this video we look at strong vs weak bases and how they dissociate in water. Strong bases will fully dissociate (completely separate into cations and anions) while weak bases will have only a little dissociation. This means that solutions with strong bases are strong electrolytes because there are a lot of ions in the solution. Weak bases ...








